The Renin-Angiotensin-Aldosterone System (RAAS): A Step-by-Step Guide
The renin-angiotensin-aldosterone system is a crucial hormonal cascade that regulates blood pressure, fluid balance, and vascular resistance. Here’s a comprehensive breakdown of this pathway:
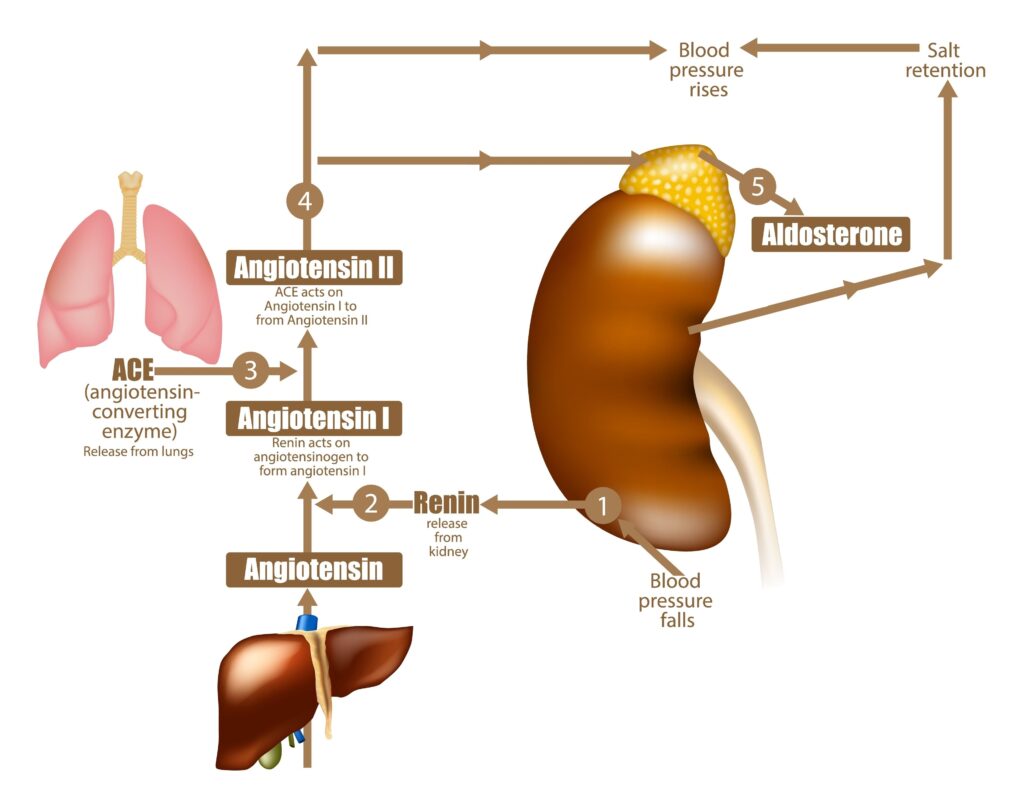
Step 1: Stimulus Detection
The pathway begins when specialized cells in the kidneys (juxtaglomerular cells) detect one of three key stimuli:
- Decreased arterial blood pressure (detected by baroreceptors)
- Decreased sodium chloride levels in the distal tubule (detected by macula densa cells)
- Sympathetic nervous system activation via ?1-adrenergic receptors
Step 2: Renin Release
In response to these stimuli, juxtaglomerular cells secrete renin, a proteolytic enzyme, into the bloodstream. Renin is the rate-limiting factor in the RAAS cascade, making its release a critical regulatory step.
Step 3: Angiotensinogen to Angiotensin I Conversion
Renin acts on angiotensinogen, a protein produced primarily by the liver. Angiotensinogen is an ?2-globulin with 452 amino acids. Renin cleaves angiotensinogen at the Leu10-Val11 bond, releasing the decapeptide angiotensin I (Ang I).
Step 4: Angiotensin I to Angiotensin II Conversion
Angiotensin I is relatively inactive and serves primarily as a precursor. It circulates until it encounters angiotensin-converting enzyme (ACE), which is abundant in pulmonary capillary endothelium. ACE cleaves two amino acids from the C-terminus of Ang I, converting it to the octapeptide angiotensin II (Ang II).
Step 5: Angiotensin II Action
Ang II is the primary effector molecule of the RAAS with a half-life of only 1-2 minutes. It exerts its effects by binding to two main receptors:
AT1 Receptor Effects:
- Vasoconstriction: Causes contraction of vascular smooth muscle, particularly in arterioles
- Aldosterone synthesis: Stimulates the adrenal cortex to produce aldosterone
- ADH (vasopressin) release: Enhances water reabsorption via the posterior pituitary
- Thirst stimulation: Acts on the hypothalamus to increase water intake
- Sympathetic potentiation: Enhances norepinephrine release and inhibits reuptake
- Cardiac effects: Promotes cardiac hypertrophy and increased contractility
- Renal effects: Increases sodium reabsorption in proximal tubules
AT2 Receptor Effects:
- Generally opposes AT1 effects, including vasodilation and natriuresis
- Involved in tissue growth, development, and repair
Step 6: Aldosterone Production
Ang II stimulates the zona glomerulosa of the adrenal cortex to synthesize and release aldosterone, a mineralocorticoid hormone.
Step 7: Aldosterone Action
Aldosterone acts on mineralocorticoid receptors in the distal tubules and collecting ducts of the kidneys to:
- Increase expression of the Na?/K?-ATPase pump at the basolateral membrane
- Upregulate epithelial sodium channels (ENaC) at the apical membrane
- Enhance sodium reabsorption and potassium secretion
- Promote water reabsorption following sodium (osmotic effect)
Step 8: Blood Pressure Regulation
The combined effects of this cascade lead to:
- Increased total peripheral resistance via vasoconstriction
- Increased cardiac output via direct cardiac effects and increased preload
- Increased blood volume via sodium and water retention
- Net result: Elevation of blood pressure
Step 9: Negative Feedback
The elevated blood pressure provides negative feedback to the juxtaglomerular apparatus, inhibiting further renin release and establishing homeostatic control.
Alternative Pathways and Modulators
- ACE2: Converts Ang II to Ang(1-7), which has vasodilatory and anti-inflammatory effects via the Mas receptor
- Chymase pathway: Non-ACE production of Ang II, particularly in tissue-specific RAAS
- Aldosterone escape: Mechanism that limits excessive sodium retention
- Natriuretic peptides (ANP, BNP): Counter-regulatory hormones that antagonize RAAS effects
Clinical Significance
Understanding this pathway has led to the development of several drug classes:
- ACE inhibitors (e.g., enalapril, lisinopril)
- Angiotensin II receptor blockers (e.g., losartan, valsartan)
- Direct renin inhibitors (e.g., aliskiren)
- Aldosterone antagonists (e.g., spironolactone, eplerenone)
These medications are cornerstones in treating hypertension, heart failure, kidney disease, and diabetes-related complications.
References
- Forrester SJ, Booz GW, Sigmund CD, Coffman TM, Kawai T, Rizzo V, Scalia R, Eguchi S. Angiotensin II signal transduction: An update on mechanisms of physiology and pathophysiology. Physiol Rev. 2022;102(2):1037-1104.
- South AM, Tomlinson L, Edmonston D, Hiremath S, Sparks MA. Controversies of renin-angiotensin system inhibition during the COVID-19 pandemic. Nat Rev Nephrol. 2022;18(3):166-180.
- Riccioni ME, Bizzarri B, Mazza M, Tosoni A, Birocchi E. The renin-angiotensin-aldosterone system: new insights into chronic kidney disease. Int J Mol Sci. 2023;24(1):422.
- Guzik TJ, Mohiddin SA, Dimarco A, Patel V, Savvatis K, Marelli-Berg FM, et al. COVID-19 and the cardiovascular system: implications for risk assessment, diagnosis, and treatment options. Cardiovasc Res. 2022;118(3):752-769.
- Jackson CB, Farzan M, Chen B, Choe H. Mechanisms of SARS-CoV-2 entry into cells. Nat Rev Mol Cell Biol. 2022;23(1):3-20.

Nancy L. Kimmel obtained her PhD in Environmental Engineering in 2002, then went on to teach Physics and Mechanical Engineering at Lawrence Technological University, Henry Ford College and Oakland University. She obtained her Associate in Nursing from Henry Ford College and then went on to earn her Master Degree as a Family Nurse Practitioner and became Board Certified working as a licensed FNP in the State of Michigan. She then went on to Medical School where she is now in her 3rd year, and is also in the process of obtaining her Doctorate in Nursing Practice through Chamberlin University. She has authored the NET Study Guide, as well a several books on subjects of Math, ECG/EKG and Phlebotomy. She holds a patent on an Air Filter through the U.S. Patent Office.
313-826-2381

